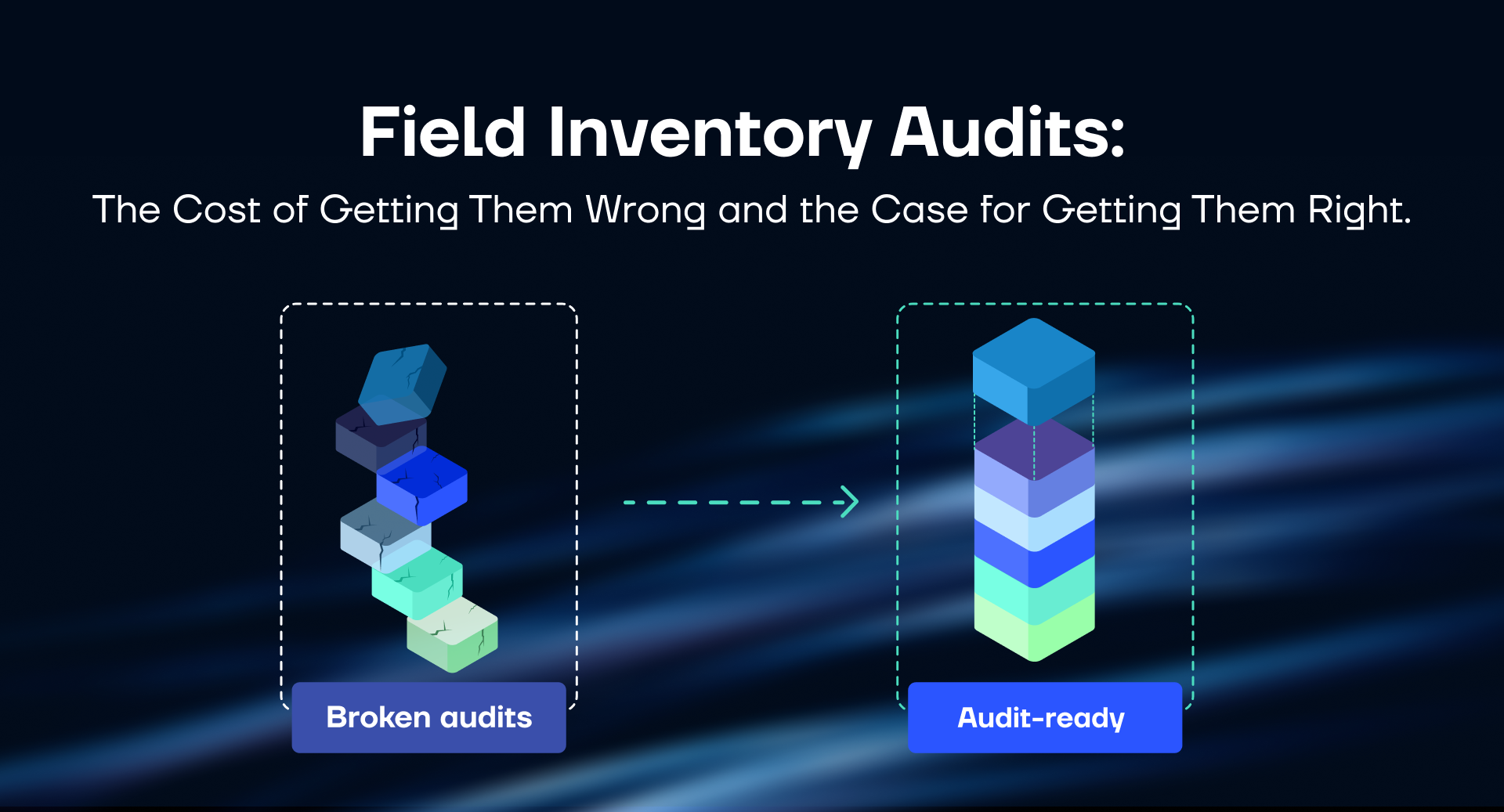
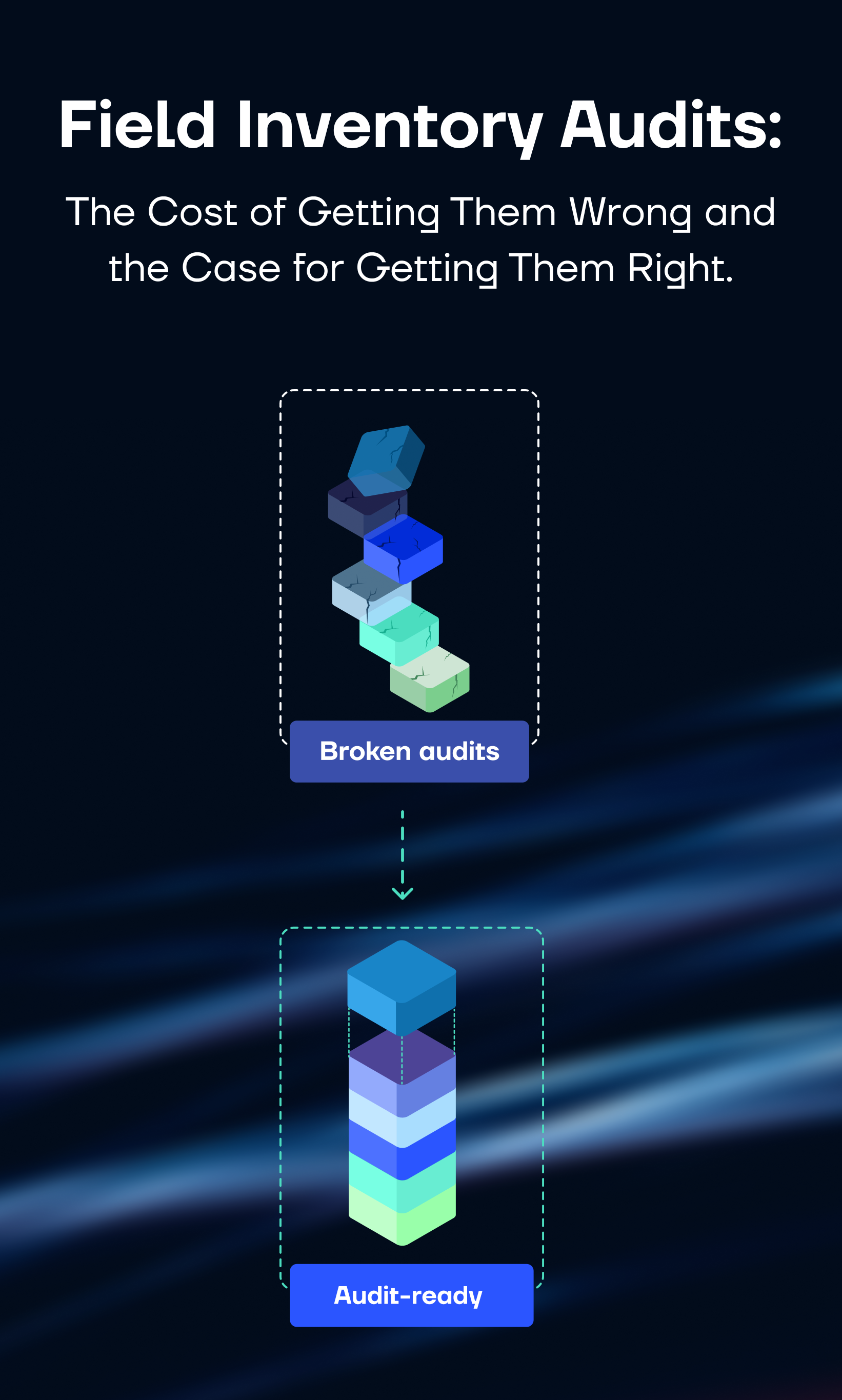
Most MedTech operations leaders know their audit process isn't great. Field reps scramble at month-end. Reconciliation takes days. The numbers that come out don't match the ERP — and everyone moves on until next month.
What rarely gets calculated is the full financial weight of that pattern, or what it would actually take to fix it.
This article covers both. First, what a broken audit process costs — in working capital, labor, and compliance exposure. Second, a diagnostic of where audit processes typically break down, across six operational dimensions, and what closing each gap actually returns. The goal is a clear picture of where your organization stands and what the investment case looks like.
The audit problem is not unsolvable. It requires an honest look at where the gaps run deepest — across data, tools, ownership, and integration — and investment in the right places to fix them.
Where the Financial Exposure Lives
Working Capital
A MedTech company with $500M in revenue can easily have $100M or more tied up in field inventory — consignment product, surgical trays, and trunk stock distributed across hundreds of field reps. When audit processes can't produce an accurate picture of what products are where, that number grows, unchecked. Procurement over-orders because the field looks short. Reps hoard more inventory than they need because no one's asking for it back. Product expires in the field because no one tracked it to a location with enough lead time to intervene.
Each of those outcomes has a number attached to it. Organizations that have modernized their audit infrastructure routinely report reductions in field inventory carrying costs of 20–40%, along with significant drops in expired inventory. In absolute terms, at scale, those are eight-figure recoveries.
Labor and Rep Productivity
When audit processes depend on manual coordination — office staff building spreadsheets, reps counting product against paper logs, reconciliation done by hand in the ERP — the labor hours add up fast. More importantly, they're the wrong labor hours. Your field reps find themselves doing cycle counts instead of being in the OR. Every hour spent on manual auditing is an hour not spent in front of a surgeon, not closing a case, not building the relationships that drive revenue.
Compliance and Regulatory Exposure
As FDA oversight of field inventory tightens and MDR obligations expand, the documentation gaps left by fragmented audit processes become more than operationally inconvenient. They carry real regulatory risk. The organizations that discover this after an audit failure pay a much higher price than those who build the right infrastructure beforehand.
Six Dimensions: Where Audits Break Down and What Fixing Them Returns
Most organizations respond to audit failures by adding more process: more reminders, more spreadsheets, more manual checkpoints. And it never works. This is because the gaps aren't just process gaps. They run across data quality, tooling, ownership, and system integration. Closing them requires understanding where each one lives and what it costs to leave it open.
1. Inventory Visibility
The gap: Without accurate, accessible inventory balances across field locations, every downstream audit activity is a guessing exercise. Data exists in most organizations, but it's fragmented across systems, updated infrequently, and impossible to trust at the moment it's needed most. ERP records reflect what left the warehouse — they don't know a rep transferred half a tray to a colleague last Tuesday, or that trunk stock has been sitting in a car for three months.
What good looks like: Real-time, system-driven visibility across every consignment location and lot level — without manual input. Teams can answer "where is our inventory right now?" with confidence, not approximation.
What it returns: Accurate visibility is the foundation that makes every other audit improvement possible. Without it, organizations over-order, carry excess safety stock, and absorb expiration losses that could have been prevented. With it, field inventory carrying costs drop 20–40% and expired inventory losses fall sharply.
2. Scanning Tools
The gap: Paper-based audits and spreadsheet counts are still common across the industry. They're slow, error-prone, and guarantee that someone spends hours cleaning up what should have been captured correctly the first time. Mobile scanning improves speed, but only if it syncs with systems of record in real time — disconnected mobile tools just move the manual reconciliation problem downstream. There's also a more basic infrastructure problem: many scanning tools require an internet connection to function, and a significant share of field audits happen in hospital supply rooms, basement storage areas, and sterile processing departments where connectivity is unreliable or nonexistent. When the tool stops working mid-count, reps fall back to paper — which defeats the purpose entirely.
What good looks like: Integrated mobile and desktop systems that sync with approvals and structured reconciliation workflows. At the highest maturity level, RFID and automated scanning deliver continuous visibility with near-zero manual auditing burden.
What it returns: Cleaner data captured at the point of count means less reconciliation labor, fewer discrepancies, and audit cycles that take hours instead of days. The productivity return compounds across every rep and every audit period.
3. Planning Audits
The gap: When reps find out about audits through an email from the home office, completion rates are unpredictable and documentation is inconsistent. Missed audits don't just create operational gaps — they create regulatory ones. Manual scheduling also means that as territory complexity grows, planning becomes a bottleneck the ops team can't scale.
What good looks like: Audits automatically generated, assigned, and tracked — with centralized status visibility and rep notifications built in. Compliance becomes a managed metric, not a monthly fire drill.
What it returns: Higher completion rates, consistent documentation, and a material reduction in the labor cost of audit coordination. Organizations that automate audit planning also reduce the compliance exposure that accumulates when audits are missed or poorly documented.
4. Organization and Team
The gap: In most organizations, audit ownership sits entirely with individual field reps — which means audit quality varies by rep. There's no cross-functional accountability, no defined escalation path when something's wrong, and no structural connection between the audit outcome and the ops team that needs to act on it.
What good looks like: Audit ownership distributed across sales and operations, with defined roles, shared accountability, and audit processes embedded into standard workflows rather than treated as a separate activity that sales tolerates.
What it returns: Consistency. When audit quality doesn't depend on which rep is running the count, organizations get data they can actually act on. Cross-functional ownership also accelerates reconciliation — issues get resolved faster when ops has visibility and authority rather than waiting on a rep to follow up.
5. Integration of Tools
The gap: An audit process that doesn't connect to the ERP generates compliance paperwork, not operational improvement. Data captured in the field stays in the field — it doesn't update inventory records, trigger adjustments, or feed the financial systems that leadership relies on. The audit happened, but the system of record didn't change.
What good looks like: Reconciled audit items initiate ERP transactions automatically. Sub-location tracking, inventory adjustments, and approval workflows execute without manual entry. The audit becomes a feedback loop that keeps the system of record accurate in real time.
What it returns: Elimination of the manual ERP update burden, significantly higher data accuracy, and a direct connection between field audit activity and financial reporting. Organizations at this maturity level don't have a "field inventory number" and a "real number" — they have one number.
6. Reconciliation
The gap: Counting inventory is the easy part. Ensuring that what was counted is accurately reflected in the ERP — with discrepancies resolved, adjustments made, and outcomes documented — is where manual processes collapse under their own weight. Reconciliation is often the last step, the most labor-intensive, and the most likely to be done inconsistently or skipped entirely under time pressure.
What good looks like: Automated reconciliation that resolves common scenarios across reps, locations, and item types without manual intervention. Exceptions are flagged and escalated; routine adjustments happen automatically.
What it returns: Books that are actually reliable. Audit labor costs fall sharply when reconciliation is automated. More importantly, the financial reporting that flows from field inventory data becomes trustworthy — which changes how confidently leadership can make inventory investment and working capital decisions.
Where to Start
The organizations that modernize audit operations most successfully don't try to close all six gaps at once. They start with an honest assessment of where they are across each dimension — visibility, scanning tools, planning, organization, integration, and reconciliation — and prioritize the gaps driving the most cost.
For most, inventory visibility is the right starting point. Without accurate, accessible data on what's in the field and where, improvements to scanning tools, planning workflows, or reconciliation processes are constrained. Clean data is the foundation everything else is built on.
Movemedical's field inventory platform addresses all six dimensions — from real-time visibility across consignment locations and lot levels to automated audit assignment, mobile scanning, and ERP-integrated reconciliation. Leading MedTech organizations running complex surgical rep models at scale have used Movemedical to convert their audit process from a quarterly liability into a continuous operational advantage.
The audit problem is solvable. The organizations that solve it treat it as an infrastructure investment, not a process fix — and they measure the return in working capital recovered, inventory losses eliminated, and selling time reclaimed.
Ready to assess where your organization stands across the six dimensions of audit readiness? Take Movemedical's Audit Readiness Scorecard — a two-minute assessment that maps your current capabilities and delivers a customized executive report with industry benchmarks.
Take the Audit Readiness Scorecard →

















.png)



.svg)
.svg)
.svg)
.svg)


